My current doc: Josepa Rigau Av Catalunya, 12, 3º, 1ª 43002 Tarragona Spain +34977220358 (I do recommend! hoeopathy and biological medicine, significant improvement)
My previous docs: De Meirleir (www.redlabs.be), Dra Quintana (CMD), (Lots of medication, antibiotics etc... no significant improvement)
What is Chronic Fatigue Syndrome? (Post # 1)
We all get tired. Many of us at times have felt depressed. But the mystery known as chronic fatigue syndrome (CFS) is not like the normal ups and downs we experience in everyday life. The early sign of this illness is a strong and noticeable fatigue that comes on suddenly and often comes and goes or never stops. You feel too tired to do normal activities or are easily exhausted with no apparent reason. Unlike the mind fog of a serious hangover, to which researchers have compared CFS, the profound weakness of CFS does not go away with a few good nights of sleep. Instead, it steals your energy and vigor over months and sometimes years.
The following chart shows some statistics taken in Australia regarding length of continuing disability symptoms, although is actually very unpredictable to forecast when are you going to improve your symptoms, and also if they may come back at a later stage.
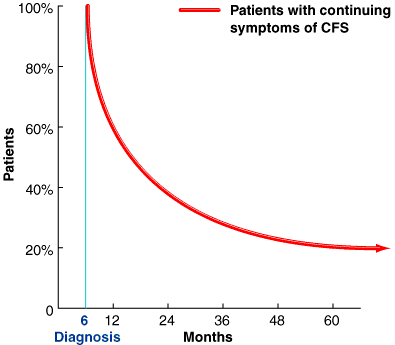
Despite of the chart of persistence of CFS symptoms, full recovery is estimated at 10 percent, with the greatest chance of recovery appearing to be within the first five years of illness. Some people cycle between periods of relatively good health and illness, and some gradually worsen over time. Others neither get worse nor better, while some improve gradually but never fully recover
How does CFS begin, and what are CFS symptoms?
For many people, CFS begins after a bout with a cold, bronchitis, hepatitis, or an intestinal bug. For some, it follows a bout of infectious mononucleosis, or mono, which temporarily saps the energy of many teenagers and young adults. Often, people say that their illnesses started during a period of high stress. In others, CFS develops more gradually, with no clear illness or other event starting it.
DEFINITION:
Just for a sake of clarity, the name of this illness has brought a lot of controversy, and currently they are trying to change it to neuroendocrineimmune dysfunction syndrome, or NDS, which better reflects the symptoms of this illness, is not just fatigue...
Chronic Fatigue Syndrome is an Illness characterized by a permanent fatigue (that it does not improve with the rest), for at least six consecutive months, accompanied of other symptoms as difficulty of concentration, lost of memory, non-refreshing sleep, muscular pains, pains you will articulate (without inflammation), migraines, general discomfort post exercise that extends more than 24 hours and alterations of the sleep.
This Chronic Fatigue Syndrome has also been called Immune Malfunction, epidemic Neuromiastenia and Myalgic Encephalomyelitis.
The following chat is a comprehensive way to measure the varied scope of this illness in terms of symptoms and effects in different parts of the body:
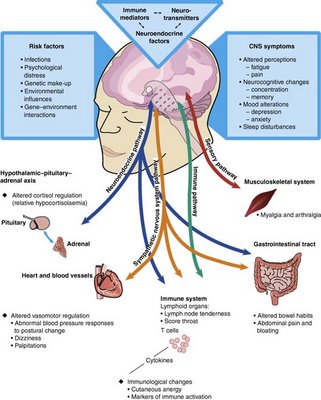
The illness has proven to be both complex and mysterious, and there is still no known cause or cure. However, there is abundant scientific evidence that CFS is a real biologic illness, not a psychiatric condition. And there are treatment options that can help patients manage symptoms, cope with the impact of the illness, improve function and manage activity levels.
CAUSES:
There is not known yet the final cause of it, but it is believed that a viral infection is involved in it. Among the viruses responsible for CFS are: Epstein-Barr, HIV, HHV-6, Cytomegalovirus, retrovirus or enterovirus.
The World Health Organization has listed the discrete disease, CFS under neurological disorders specifically excluding it from psychiatric disorders. Doctors treating patients medically diagnosed with CFS describe them are more functionally ill than cancer patients undergoing chemotherapy, patients with HIV, Type 2 diabetes and another neurological disorder MS.
In one study, Ablashi et al. (15) found that 25% of the sera &am 300 CFS patients, tested for HHV-6 IgG and EBV-VCA IgG antibody, showed elevated antibodies to both HHV-6 and EBV.
There has been considerable interest in investigating its possible role in CFS. Most of us have already been infected with the virus in our first year of life. In most individuals the virus is latent. When HHV-6 is reactivated, or during reinfection, it may contribute to CFS.
Evidence of the involvement of HHV-6 in CFS, compared to that of other human herpes viruses (EBV, CMV, HSV-1 and 2, VZV, HHV-7), is much stronger. The evidence is based on:
1. Elevated IgG antibody;
2. Detection of anti-IgM antibody in equal to or less than 50% of patients, which is a good indication of virus reactivation;
3. Detection of HHV-6 antigen expressing cells in the peripheral blood mononuclear cells of CFS patients by culture techniques;
4. Detection of HHV-6 DNA in lymphocytes of CFS patients by PCR and Southern blot hybridization (22-23,33,35-36).
DIAGNOSTIC AND BIOLOGICAL MARKER:
In the following chart, we can see how clinical diagnostic can be made, and RNASe biological marker is simply a confirmation of the clinical diagnostic.
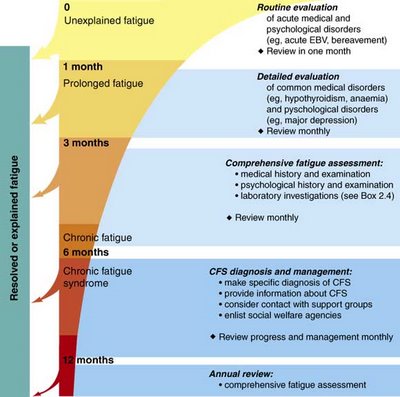
Quantitative Measurement of RNase L Proteins: The Confirmatory Test to Aid in the Diagnosis of Chronic Fatigue Syndrome
Chronic Fatigue Syndrome (CFS) is recognized as one of the most common chronic illnesses in the world. CFS is a complex disease syndrome of unknown etiology, afflicting people of all ages. Currently CFS is defined by its symptoms (1), the hallmark of which is chronic, debilitating fatigue of six months or more in duration. In addition, patients suffer from a number of physical problems including myalgia, arthralgia, cognitive impairment, and sleep disorders.
Further research is needed to develop a standardized diagnostic test for ME/CFS. For example, a test that shows great potential as a blood marker is the determination of the ratio of normal 80 kDa RNase L, to the low molecular weight 37 kDa RNase L found in ME/CFS patients. This has been shown to accurately distinguish ME/CFS patients from healthy controls.
This as well as other promising candidates for a standardized diagnostic test for ME/CFS warrants further research. It would be helpful if research studies distinguished between mild and severe cases, and also between newly diagnosed cases and those in chronic stages of ME/CFS.
Emergence of a Possible Diagnostic Marker
In 1995, Dr. Robert Suhadolnik and his co-workers at Temple University, Philadelphia, PA, detected a novel intracellular protein related to RNase L, one of the interferon-inducible enzymes. These enzymes, which also include 2'-5' Oligoadenylate Synthetase, and double-stranded RNA dependent Protein Kinase, play a key role in protecting the cell from viral infection. (2,3) The novel protein was determined to have a molecular weight of approximately half of the native RNase L (and is thus referred to as the Low Molecular Weight RNase L or 'LMW,' while the native RNase L protein is referred to as the High Molecular Weight (HMW) species).
Working together with clinicians Daniel Peterson and Paul Cheney, Dr. Suhadolnik was able to demonstrate the presence of this LMW protein in a subset of patients with CFS. (4,5) His findings were independently confirmed by Dr. Bernard Lebleu, at the Institute for Genetic Molecular Medicine, Montpellier University, Montpellier, France, working together with clinician Kenny De Meirleir at the Free University of Brussels, Belgium. (6)
Development of a Clinical Assay
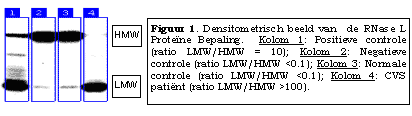
In 1998, R.E.D. Laboratories began offering the assay for the RNase L LMW protein. The assay is performed by 1) preparation of a cytoplasm extract of the patient's peripheral blood mononuclear cells, 2) combination of this extract with a labeled probe that binds specifically to 2'-5'A-binding proteins such as RNase L and the LMW species, 3) SDS-polyacrylamide gel electrophoresis, and 4) densitometry to determine the relative quantities of 2'-5'A-binding proteins (see Figure 1 below).
Results & Interpretation
Results are quantified with normal reference ranges provided. A numerical value is calculated by densitometry as "the amount of LMW protein present divided by the amount of native (HMW) RNase L present" multiplied by a factor of 10 (or (LMW/HMW) x 10).
Preliminary data indicate the following:
Negative: Ratio <0.1 - 1.9 XXXXX Positive: Ratio = 2.0 or more
(Please note: The results of this test should be used in addition to all other relevant clinical data before making a diagnosis and/or a recommendation for treatment. Ranges are subject to change dependent on future data.)
The biological scoreboards that can have a big utility in the diagnosis, stratification of the severity of the fatigue, and later in the treatment, it would be the quotient Rnasa L (37 kDa/83 kDa), activity Rnasa L, elastasa would monoquote and activity of the PKR, which they can already determine in laboratories of investigation of Europe.
In relation to the implication of these scoreboards in the response of the treatment of the SFC with the negativization of Rnasa L, the clinical therapeutic essays are of interest realized with Pli-1, 12-CU (ampligen), that Rnasa L is possible to negativate, and that contributes with the progress of the clinical Symptomatology in the SFC.
WHY EARLY DIAGNOSIS IS SO IMPORTANT: THE PATIENT EXPERIENCE
Imagine being a fit and healthy person who picks up a simple viral infection but fails to return to a normal pattern of health. You develop a classic range of ME/CFS symptoms: exhaustion after minimal amounts of activity, severe problems with memory and concentration, and other disabling neurological symptoms. Your doctor is either dismissive of your symptoms, or is unable to provide a satisfactory explanation as to what is going wrong, and so fails to offer any form of effective management advice.
This situation is all too common. Diagnostic delay creates frustration, even despair, because of the resulting lack of appropriate management advice in the crucial early stages. Worse still, people can be given totally inappropriate and harmful recommendations on management, especially in relation to striking the correct balance between activity and rest.
Organizing dealings with family, friends, employers or schools becomes very difficult. One common consequence is that people in the very early stages of ME/CFS are told to return to work or school far too quickly, instead of benefiting from a period of convalescence followed by a gradual and flexible attempt at returning to education or occupational activities.
State and private sickness benefits can be very difficult to obtain in the absence of a clear diagnostic opinion that can be written on a sick note. As a consequence, people are denied the appropriate sickness benefits, practical help with their care or mobility needs, and access to social services.
TREATMENTS:
Unfortunately, the only treatments available are symptomatic, and there is not yet a cure for CFS. This is a progressive illness, and the prognosis for recovery varies from case to case. Actually only 10% recover completely, and the rest simply reduce the severity of the symptoms and can have a "normal" life after 1, 2, 5 or 10 years. There are around 25% of patients that show a high severity of symptoms and may live in a wheel chair or bed bound permanently.
Nevertheless, is very relevant to have an early diagnostic and treatment, because otherwise we will probably too many efforts. Energy managent and proper treatment can avoid relapsing in more severe symptoms. The existing treatments for this illness are based on trial and error, because every patient reacts differently.
The Vitamin C and the N-Acetil-Cisteína are standard approaches agreed in the treatment for CFS symptoms.
Ampligen: The drug polyl:polyC12U (Ampligen) is a form of genetic material called dsRNA. It inhibits viral replication. While early studies yielded questionable results, more carefully designed, larger studies are now showing clear benefits to CFS patients. The largest study to date, presented in 2004, found that compared to placebo, Ampligen considerably increased exercise ability and caused no serious adverse effects. At this time Ampligen must be administered by intravenous injection and is extremely expensive. If it becomes available in pill form, it may gain insurance reimbursement and wider use among patients.
Ampligen is an immune stimulator and viral modulator. Ampligen is available for moderate to severely ill patients who have been following other treatments and have RNase L abnormalities. Ampligen is a mismatched ds RNA, which induces the interferon/2-5 synthetase pathway, and controls immune deregulation, possibly caused by an abnormal RNase L enzyme. It may inhibit viral attachment to cellular receptors and/ or inhibit intracellular maturation of the virus. Initial studies have shown that a baseline 24-week treatment produces positive results, continuing to several years. Significant improvements were in exercise capacity, memory and cognitive ability, activity of daily living and fewer emergency room visits and hospitalizations, as compared to controls. Possible side effects–flu-like symptoms, myalgia, headaches at initiation (later abated), lightheadedness and facial flushing. RCTs continue.
The positive effect of Ampligen is that it regulars the abnormally high RNASE showed by CFS patients, and reduces the CFS symptoms, because of this regulation of the main anti viral response system. The treatment is not suitable with patients with an elevated PKR, because Ampligen, could increase even more this value and would have an impact on the severity of the symptoms. When this is the case, the use of certain antibiotics can be appropriate. (See more on Ampligen in the post dedicated to Ampligen)
Azithromycin: Some recognized doctors as expert in CFS think that the treatment with certain antibiotics essentially of the group of the beta - lactámics, can be useful in cases of increased elastasas, since precisely they have an inhibitory role. Detection oh abnormally high elastase indicates inflammation related to an active infection, when we reduce elastase, we also reduce inflammation, pain, fatigue and other symptoms. All the experts in SFC do not share this opinion.
A number of antibiotics are being investigated for confirmed infections of mycoplasma or Chlamydia. As the rationale and dosage protocols for these treatments are still under investigation, it is premature to recommend this treatment. A brief description is included for your information. The antibiotics being investigated include doxycycline (100 mg bid-tid), clarithromycin (750-100 mg daily on a q12h schedule clarithromycin (750-100 mg daily on a q12h schedule), ciprofloxacin (750 mg bid), and azithromycin (500 mg as a single dose). Generally the trials are for 6 months or until improvement is noted, then multiple courses of 6 weeks on followed by a two-week break. Normal gut flora should be replaced and immune boosters are recommended.
Probiotics: VSL3, Acidophilus, Ultra Levura... this is useful to reestablish the intestinal flora, especially if antibiotic treatment has taken place.
Carnitine: Some experts recommend the use of carnitine, with a high effectiveness on improvement of symptoms after 4 months of treatment. Unfortunately, this is quite expensive treatment, and the moment it is interrupted, the symptoms come back.
Vitamin B12 is used to boost the immune system and provide energy. All the experts generally accept its use. Is easier to use the sublingual format of 1.000mcg a day or even 10.000 mcg a day. When used as intramuscular injection the format is 100mcg every week.
Valganciclovir: This is a drug used to treat herpes infections has produced a dramatic improvement in patients severely affected by ME. Sufferers who for years had been unable to leave their homes now report being able to resume normal life. The results, reported at a scientific conference in June 2006 by Professor Jose Montoya of Stanford University in California, involved 12 patients who had been given the powerful drug valganciclovir, which targets the human herpes virus (HHV-6). Nine of the patients experienced a great improvement. Nevertheless this drug may have dangerous side effects, and still needs to be further investigated. It is presently being investigated by double-blinded placebo-controlled randomized trial studies
Valanciclovir: Similarly, this is an Herpes antiviral, with much less risks in terms of side effects, which are used by some experts to treat chronic EBV viral infections. Nevertheless EBV is not a treatable Herpes virus as it is Heroes Zoster or Herpes Simplex. The logic behind this treatment is that instead of using it for 3 or 4 days, the treatment is prolonged during 14 days in order to achieve an effect on EBV. The efficacy varies depends on the patient.
LINKS:
I included in the right side several links that I find useful for understanding this illness and also sites that help you to proceed in case you have CFS and need legal advice in case your employer is making things difficult for you. The worst part of this illness is many times the fact that you have to convince the rest that you are ill, even when your face doesn't show it. It is quite frustrating to be ill, and to face that the ignorant put under question the severity of your symptoms.
1 comment:
Great blog!
For people in the Netherlands wo want to know more about the ideas and work of dr. de Meirleir
see http://www.hetalternatief.org.
Post a Comment